Small nucleic acid-based treatments are rapidly emerging as a significant advancement in the medical field, continuing the paths of small-molecule medications and antibody-based biologics.
These medications are not similar to the standard drugs which work on proteins. Instead, they are short, chemically made nucleic acid strands that only target disease-related mRNA or non-coding RNAs. By directly affecting the gene-expression pathway, from DNA to RNA to pathway, they provide precision, modularity, and versatility that prior treatments could not match. This change in mechanism has made oligonucleotide-based medicines a revolutionary new area of modern medicine.
Inclisiran is a groundbreaking new drug among these revolutionary prospects. This is the first siRNA treatment in the world to be authorized for lipid management, marking a new era in RNA-based medicines. Inclisiran uses RNA interference (RNAi) technology to turn off the PCSK9 gene in the liver. This stops the PCSK9 protein from being made, which lowers LDL cholesterol levels by a large amount. For people who are at risk of heart disease, this long-acting, twice-a-year dose alternative is both easy to use and has strong clinical results.

Figure 1: Inclisiran Sodium Injection
Inclisiran is composed of two chemically produced oligonucleotide strands: a 21-mer sense strand modified with a GalNAc ligand for hepatocyte targeting and a complementary antisense strand. Together, they bind to and degrade PCSK9 mRNA, resulting in prolonged gene silence. Because of this intricacy, its manufacturing relies substantially on modern oligo synthesis processes, rigorous process control, and high-precision equipment.
This case study analyzes the whole lab-scale production method of Inclisiran utilizing the Bio Oligo 100 laboratory nucleic acid synthesis Instrument,. By merging solid-phase chemical synthesis, real-time monitoring, precision engineering, and optimal software control, the workflow demonstrates how contemporary oligonucleotide synthesizer platforms facilitate the effective production of complex siRNA therapies.
Why Inclisiran Manufacturing Requires High-Precision Oligo Engineering?
The synthesis of Inclisiran demands continuous coupling efficiency, excellent reagent control, and low impurity production. Each nucleotide addition is a multi-step chemical cycle comprising deprotection, coupling, oxidation or sulfurization, capping, and washing. Any deviation, whether owing to reagent quality, flow rate, or reaction time, can lead to truncation products (N-1), extended products (N+1), or depurination.
Therefore, a reliable and intelligent oligo synthesizer instrument is vital for assure that every monomer is precisely introduced to the expanding chain. Lab-grade systems like the Bio-Oligo series are designed to fulfill this demand by delivering programmable automation, real-time monitoring, and fine-tuned control across every synthesis cycle.
Introduction to the Bio Oligo 100 System
The Bio Oligo 100 is a laboratory-scale oligonucleotide synthesizer equipment designed for DNA and RNA synthesis ranging from 50 pmol to 9 mmol. Built with modular architecture and an advanced valve-switching control system, it allows varied applications, from basic R&D sequences to therapeutic-grade complicated structures.

Figure 2: Bio Oligo 100 Laboratory Nucleic Acid Synthesizer
Key features aiding Inclisiran synthesis include:
- High-precision piston pumps providing consistent reagent delivery
- A flow-through solid-phase system optimized for linear scalability and decreased reagent usage
- Intelligent workstation software allowing mid-run adjustments
- Real-time UV detection for deprotection monitoring
- Conductivity monitoring for coupling, oxidation, and washing stages
- Compatibility with multiple column sizes for small-scale and large-scale oligo synthesis
These features make the technology perfect for custom oligonucleotide synthesis, synthetic oligos, and the generation of highly specialized double-stranded RNA sequences such as Inclisiran.
| Step | Flow Rate |
| Deprotection | 400 cm/h |
| Deprotection Wash | 200 + 200 cm/h |
| Coupling Injection | 2.5 + 3.75 ml/min |
| Coupling | 230 cm/h |
| Coupling Wash | 200 + 200 cm/h |
| Oxidation | 3.25 ml/min |
| Oxidation Wash | 200 + 200 cm/h |
| Sulfurization | 1.39 ml/min |
| Sulfurization Wash | 200 + 200 cm/h |
| Capping | 0.5 + 0.5 cv/min |
| Capping Wash | 200 + 200 cm/h |
| DEA (Cleavage) | 75 cm/h |
| Wash | 200 + 200 cm/h |
Table 1: Process Parameters for Inclisiran Synthesis on the Bio Oligo 100 System
(*Process description for a 6.3 mL synthesis column; specific parameters are programmed into the software workstation.)
Chronological Walkthrough of Inclisiran Synthesis Using Bio Oligo 100
1. Column Preparation & Reagent Loading
The reaction was performed in a 6.3mL synthesis column. The system was connected with reagents such as monomer phosphoramidites, activators, oxidizing agents, sulfurizing agents, and capping solutions. The software interface automatically estimates predicted consumption and in real time to make sure sufficient quantity of material is
available.
2. Deprotection Monitoring
- The deprotection stage commences each synthesis cycle by removing the DMT protective group.
- Using the system’s built-in UV detector, absorption values were monitored continually.
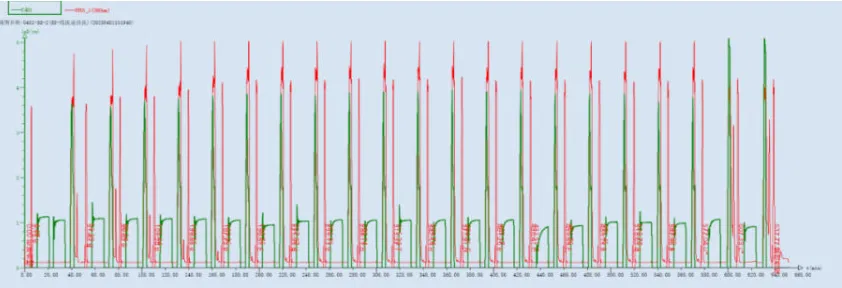
Figure 3: UV 350 nm and Conductivity Monitoring Profile During the Entire Sense Strand Synthesis Process
- A consistent decline in DMT signal showed successful deprotection, while the deprotection wash ensured total elimination of the liberated DMT.
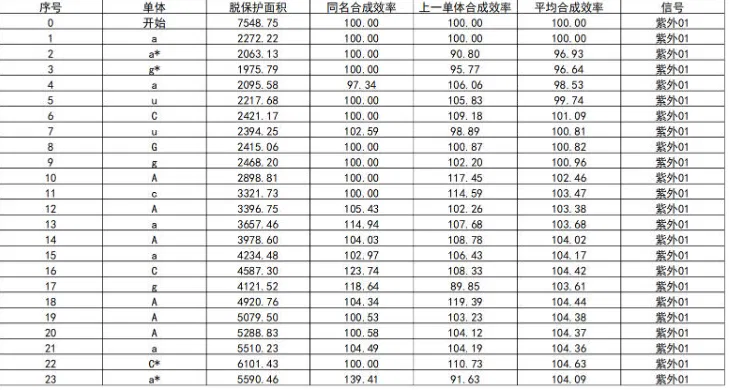
Table 3: Real-Time Monitoring of Synthesis Efficiency During Antisense Strand Synthesis
- This real-time data prevented concerns like over-deprotection, which could lead to depurination and increase unwanted side contaminants.
3. Coupling Efficiency Control
- During the coupling process, conductivity changes were evaluated to validate monomer activation and delivery.
- Any abnormal drop in conductivity alerted operators to potential reagent flow issues or sealing defects in the system.
- The automated oligonucleotide synthesis ensured tight control of monomer equivalents, considerably enhancing sequence fidelity.
4. Oxidation / Sulfurization Process
- Inclisiran’s mixed backbone chemistry needed precision switching between oxidation and sulfurization cycles.
- The system monitored the conductivity curve to certify reagent exchange completion. Only when the conductivity went below 0.05 mS/cm did the system change to the next stage, guaranteeing no oxidant remnant remained that could cause over-oxidation.
5. Cleavage & Final Wash
- After chain elongation was complete, the DEA cleavage step liberated the oligo from the solid-phase support.
- The sequence was then treated to a series of washes to eliminate remaining contaminants, ready it for downstream analysis.
Results: Crude Product Quality & Analytical Validation

Figure 7: HPLC Analysis of the Antisense Strand
HPLC study of crude Inclisiran strands indicated outstanding synthesis performance:
Sense strand crude purity: 86.2%
Antisense strand crude purity: 84.4%
Average monomer coupling efficiency: 99%

Figure 8: Mass Spectrometry Analysis of the Antisense Strand
These numbers suggest outstanding capabilities for creating high-purity full-length products prior to any oligonucleotide purification processes.
Mass spectrometry confirmed the exact predicted molecular weights for both strands, proving successful chemical alteration, proper ligand attachment, and the absence of degradation or sequence mismatch.
This outcome indicates that the DNA oligo synthesizer platform enables reliable, reproducible synthesis suitable for medicinal research objectives.
Why Bio Oligo 100 Strengthens Intech’s Nucleic Acid Workflow
By integrating the Bio-Oligo Laboratory Nucleic Acid Synthesis Instrument into Intech’s workflow, researchers gain:
- High-efficiency solid-phase synthesis
- Flexible protocol customization
- Automated reagent usage tracking
- Strong sequence accuracy for custom oligos
- Compatibility with varied DNA/RNA synthesis requirements
- Reliable scale-up possibilities with Intech – Hanbon bigger oligonucleotide synthesis machine lineup
Intech – Hanbon ‘s ecosystem covering synthesis, chromatography, preparative HPLC, and TFF supports the complete solution of oligonucleotide synthesis, from discovery to manufacture.
Conclusion
This case study shows how the Bio Oligo 100 platform is used to synthesize Inclisiran, a complex siRNA therapy, with high crude purity, remarkable coupling efficiency, and excellent reproducibility. The system is a crucial oligo synthesizer for research and early-stage drug discovery because of its intelligent automation, precise engineering, and real-time process visibility.
The platform’s capacity to provide scalable, high-performance oligonucleotide production solutions is demonstrated by Intech’s use of this reliable synthesis technique, opening the door for the development of the future of genetic medications.

Figure 9: Integrated Solution for Nucleic Acid Drug Production

