What is a Pharmaceutical Lyophilizer and How Does it Work?
The pharmaceutical industry relies heavily on precision equipment to ensure product quality. One such vital tool is the Pharmaceutical Lyophilizer. This device reduces solvent from a product, preserving fragile compounds. As per a report from Market Research Future, the global lyophilization market is projected to reach $4 billion by 2025. The growth highlights its essential role in drug development and manufacturing.
Dr. Emily Chen, an expert in pharmaceutical processing, emphasizes the importance of lyophilization. She states, "Lyophilizers are crucial for extending shelf life and ensuring product stability." Such insights underscore the device's significance for biopharmaceuticals and vaccines. However, not all formulations respond well to lyophilization. Challenges often arise during the process, requiring refinements and expertise.
Navigating these hurdles is critical for manufacturers. The experience gained through overcoming obstacles can drive innovation. Thus, understanding the nuances of a Pharmaceutical Lyophilizer is essential for industry advancement.

Definition and Purpose of Pharmaceutical Lyophilizers
Pharmaceutical lyophilizers are essential in the drug manufacturing process. These devices remove moisture from pharmaceutical products, preserving their effectiveness. By transforming liquid into powder, lyophilization protects sensitive compounds. This process is vital for biologics, vaccines, and antibiotics, ensuring stability and shelf life.
The lyophilization process involves freezing the product, followed by sublimation. During sublimation, ice transforms directly into vapor. This step is critical, as it prevents damage to the drug's structure. A report from Grand View Research estimates the global lyophilization market will reach over $5 billion by 2025, signaling its importance in the pharmaceutical industry.
Despite advancements, there are challenges. Variations in product formulation can affect lyophilization outcomes. Additionally, not all compounds respond well to this method. Some drugs may require optimization, leading to trial and error during the process. This complexity highlights the need for experienced professionals in the field.
The Lyophilization Process: Key Steps Explained
The lyophilization process is crucial in the pharmaceutical industry. It serves to preserve sensitive compounds through a careful series of steps. This method removes moisture from products, retaining their efficacy and stability. Initially, the product is frozen to create a solid state. This stage is vital, as improper freezing can damage the structure.
Next, the product undergoes primary drying, where vacuum pressure reduces the temperature. During this phase, ice sublimates into vapor. This should occur slowly to ensure uniform drying. After this, secondary drying eliminates residual moisture. It’s essential to monitor temperature closely. If it rises too fast, product degradation may occur.
**Tips:** Maintain a consistent environment throughout the process. Fluctuating temperatures can lead to failure. Monitor the vacuum conditions to prevent collapse. Each product has unique requirements. A one-size-fits-all approach often leads to mistakes. Understand the physical properties of your materials for better outcomes.
Pharmaceutical Lyophilization Process - Key Steps Data Visualization
Components and Mechanisms of a Pharmaceutical Lyophilizer
The pharmaceutical lyophilizer is an essential device in drug manufacturing. It removes water from pharmaceutical products by freeze-drying. This process stabilizes sensitive compounds, preserving their efficacy and shelf life. Understanding its components can help ensure optimal performance.
A lyophilizer consists of several key components. The freezing chamber allows substances to freeze. The vacuum chamber removes air and moisture. In addition, the condenser collects vapor. These parts work together to create a controlled environment. Temperature and pressure must be precisely managed. This requires skilled operators.
Tips: Regular maintenance of the lyophilizer is vital. Check seals and connections frequently. Small slips can lead to significant product loss. Training staff adequately is also essential. They should understand how each component functions and its importance.
Despite advancements, challenges remain. Variability in formulations can lead to inconsistent results. Operators need to adapt and monitor closely. It’s a continuous learning process. Keeping detailed logs of each run can help identify issues, leading to better outcomes in future batches.
What is a Pharmaceutical Lyophilizer and How Does it Work? - Components and Mechanisms of a Pharmaceutical Lyophilizer
| Component | Function | Specifications |
|---|---|---|
| Compressor | Removes moisture from the product. | Capacity: 5 - 50 m³/h |
| Refrigeration System | Maintains low temperatures for sublimation. | Cooling Range: -40°C to -80°C |
| Vacuum Chamber | Creates a low-pressure environment for freeze-drying. | Pressure Range: 0.1 - 100 mbar |
| Heat Exchangers | Facilitates heat transfer for efficient drying. | Surface Area: 1 - 10 m² |
| Control System | Monitors and regulates the lyophilization process. | Touchscreen Interface, Data Logging |
Applications of Lyophilization in the Pharmaceutical Industry

Lyophilization, or freeze-drying, has become a vital process in the pharmaceutical industry. This technique enhances the stability and shelf life of sensitive products, such as vaccines and biologics. By removing moisture, lyophilization prevents degradation and maintains the activity of fragile compounds. The process involves freezing the product and then reducing the pressure to allow ice to sublimate directly into vapor.
Many pharmaceutical products benefit from lyophilization. For example, antibiotics and hormones utilize this method for improved transportation and storage. Lyophilized forms are lightweight and easy to handle. However, not all substances fare well under these conditions. Some may require additional studies to determine optimal lyophilization parameters.
Challenges exist in validating the lyophilization process. Variations in formulations can affect the final product's stability. It’s essential to conduct rigorous testing to ensure product efficacy. Not all batches will meet expectations, leading to potential loss. Ongoing research is crucial in enhancing this technology for pharmaceutical applications.
Advantages and Challenges of Using Pharmaceutical Lyophilizers
The use of pharmaceutical lyophilizers in drug formulation offers significant advantages, but it also presents challenges. One major benefit is the preservation of heat-sensitive compounds. Research shows that around 30% of biologics can be effectively preserved through lyophilization, maintaining their efficacy and stability. This technique also enhances product shelf life, reducing waste and improving cost-efficiency.
However, the process is not without its difficulties. Achieving the desired product quality can be complex. Factors like freeze-drying time and vacuum levels influence the final product. A study found that nearly 20% of lyophilized products fail quality control due to inconsistency in moisture content. These failures can lead to costly recalls and decreased trust in pharmaceutical companies.
Tips: Regular calibration of equipment can mitigate some challenges. Ensure to follow stringent SOPs for processing. Continuous training for staff on operational best practices is also crucial. Implementing quality control measures throughout the process can prevent potential quality issues, ensuring consistent product reliability.
Related Posts
-

Top 10 Pharmaceutical Lyophilizer Models You Should Know?
-
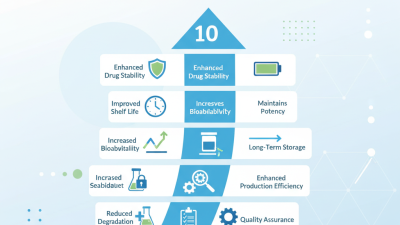
Top 10 Benefits of Using a Pharmaceutical Lyophilizer in Drug Manufacturing?
-

Essential Tips for Successful Preparative HPLC Techniques and Applications?
-

Top 5 Best Oligo Synthesizers for Precision DNA Synthesis Techniques?
-
Top Tips for High Performance Liquid Chromatography Techniques and Applications
-
Why Choosing the Right Lyophilization Equipment Manufacturers Matters?
