Top Automated Peptide Synthesis Methods and Their Applications?
Automated Peptide Synthesis represents a breakthrough in the field of biotechnology. This advanced method simplifies the production of peptides, which are vital for many applications. Researchers can now generate peptides quickly and efficiently, enhancing our understanding of biology and drug development.
Peptides play crucial roles in various biological functions. However, traditional synthesis methods are often time-consuming and complex. Automated Peptide Synthesis addresses these issues. It allows for high-throughput production and improved yield. Yet, challenges persist, such as optimizing reaction conditions and reducing errors in synthesis.
As we explore these automated methods, it is essential to reflect on their practical applications. Industries like pharmaceuticals and agriculture benefit immensely. Yet, the potential for improvement remains. We must continue to investigate ways to enhance reliability and efficiency in Automated Peptide Synthesis. This journey toward perfection is ongoing, and each step offers valuable insights.
Overview of Automated Peptide Synthesis Methods
Automated peptide synthesis methods have revolutionized the field of biotechnology. These techniques streamline the production of peptides, which are essential in pharmaceuticals and research. Conventional manual synthesis often led to inconsistencies and errors. Automation addresses these issues by enhancing precision and reproducibility.
Solid-phase peptide synthesis (SPPS) is one widely used method. In this technique, peptides are built on a solid support, allowing for easier purification. Another approach is liquid-phase synthesis, which, while less common, can yield longer peptides. Automation in these methods often involves specialized machinery that handles synthesis, monitoring reactions in real-time. Sometimes results can be unpredictable, requiring careful optimization of parameters.
While these automated processes are efficient, they are not without challenges. Issues can arise during purification or characterization phases. Sometimes, the final products exhibit undesired impurities. Researchers must continually assess and refine their methods to address these imperfections. Automation is a powerful tool, but it requires a delicate balance of technology and human oversight. More advancements are needed to enhance reliability in peptide synthesis.
Solid-Phase Peptide Synthesis: Principles and Techniques
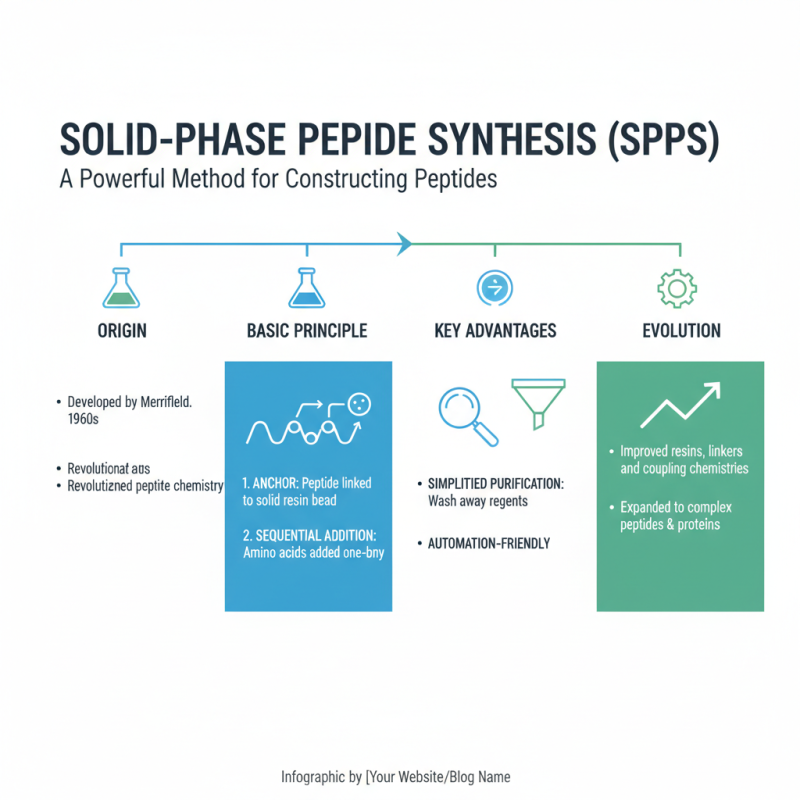
Solid-phase peptide synthesis (SPPS) is a powerful method for constructing peptides. Developed in the 1960s, it has evolved significantly. The basic principle involves anchoring a peptide onto a solid resin. This allows for the sequential addition of amino acids, simplifying purification.
During SPPS, each amino acid is added step-by-step. The process includes protecting groups to prevent unwanted reactions. These groups come with their own challenges. Sometimes, they may not fully protect the amino acids, leading to impurities. Efficient purification can become tricky.
The method is widely used in research and drug development. It enables the production of complex peptides. However, limitations exist. Not all peptides synthesize well using SPPS. Some sequences are challenging, leading to lower yields. Researchers often need to adjust protocols or explore alternative methods. Each peptide synthesis project is a learning experience, revealing both potential and challenges.
Liquid-Phase Peptide Synthesis: Methodology and Applications
Liquid-phase peptide synthesis (LPPS) stands as a prominent method in peptide production. This technique allows for the seamless assembly of amino acids into peptides. It is especially beneficial for complex peptides. According to recent industry reports, LPPS accounts for approximately 30% of the global peptide synthesis market. This method's flexibility and scalability make it preferable in research and therapeutic applications.
One of the standout features of LPPS is its ability to accommodate various protecting groups. This method also enables the synthesis of longer peptides, which can be challenging with solid-phase methods. However, working with liquid phases presents challenges. The purification process is often more complex and time-consuming. In addition, yields can vary depending on the reaction conditions.
Tip: When using LPPS, it’s essential to monitor reaction progress closely. This helps in optimizing yields and reducing by-product formation. Consideration of solvent choice and reaction temperature can have a significant impact. Don’t overlook these factors during your synthesis processes. Engaging in precise methodologies can improve outcomes dramatically. Experimenting with different conditions may lead to surprising increases in efficiency.
Top Automated Peptide Synthesis Methods and Their Applications
This chart displays the popularity of different automated peptide synthesis methods, highlighting the leading techniques used in the biochemistry field. Solid-Phase Synthesis remains the most popular method, followed closely by Liquid-Phase Synthesis.
Advancements in Automation: Robotics and Software Innovations
Recent advancements in robotics and software have transformed automated peptide synthesis. Automation significantly enhances efficiency and reliability. A recent market report suggests the peptide synthesis market is poised to reach $500 million by 2027, driven by these innovations. Robotics streamline tedious processes, reduce human error, and increase output quality. Software advancements provide smarter controls and improved data management.
However, challenges remain. Not all systems are fully integrated, which can lead to inconsistencies. Some researchers note that transitioning to automated systems might require additional training. Mistakes can occur if staff lack knowledge of both the equipment and the underlying chemistry. Emphasizing quality control processes is critical for any lab utilizing automation.
**Tip:** Consider the training needs of your team when introducing new automation technologies. Regular workshops can bridge knowledge gaps. Also, invest in software solutions that allow for user-friendly interface and data analytics. Embracing these advancements while addressing potential pitfalls ensures the benefits of automation are fully realized.
Top Automated Peptide Synthesis Methods and Their Applications
| Method | Automation Type | Applications | Key Features |
|---|---|---|---|
| Solid-Phase Peptide Synthesis (SPPS) | Fully Automated | Drug Development, Therapeutics | High-throughput, Precise Control |
| Liquid-Phase Peptide Synthesis (LPPS) | Semi-Automated | Research, Custom Peptides | Flexible, Easy Purification |
| Microfluidic Peptide Synthesis | Automated | Diagnostics, Biosensors | Low Consumption, Rapid Reaction Times |
| Automated Synthesizers with Software Integration | Fully Automated | High-Volume Production, Research | User-Friendly Interface, Data Management |
Applications of Automated Peptide Synthesis in Pharmaceutical Research
Automated peptide synthesis has become a pivotal tool in pharmaceutical research. This method allows for the rapid production of peptides, which are essential in drug development. Researchers can quickly generate large libraries of peptides for screening. This high throughput can facilitate the discovery of new therapeutic agents.
Tips: Consider the importance of quality control in peptide synthesis. Ensure the automation process is calibrated correctly. Small mistakes can lead to flawed compounds.
Pharmaceutical applications of automated peptide synthesis include vaccine development and cancer therapeutics. For example, optimizing peptide sequences can enhance immunogenicity. This process can be intricate and requires careful adjustment. Automation can streamline this task but may overlook nuances. Fine-tuning is often still necessary, and human oversight remains crucial.
Tips: Always review synthesized peptides. A quick check can reveal anomalies. Even minor inconsistencies can impact efficacy.
Related Posts
-

Essential Tips for Successful Preparative HPLC Techniques and Applications?
-

10 Essential Tips for Optimizing Biopharmaceutical Manufacturing Processes?
-

Top 10 Trends Shaping Biopharma Manufacturing Today?
-

10 Essential Tips for Optimizing Biopharma Manufacturing Efficiency
-

Digital Tips for Choosing Lyophilization Equipment Manufacturers
-

Top 5 Chromatography Systems for Efficient Laboratory Analysis?
